
Spinal Cord Injury Medicine Research Director,
TIRR Memorial Hermann
Assistant Professor, McGovern Medical School at UTHealth in Houston
At the recent National Institutes of Health conference on “SCI 2020: Launching a Decade for Disruption in SCI Research,” people with spinal cord injury (SCI) reported bladder health and function to be THEIR NUMBER ONE research priority, maintaining that No. 1 position for more than 15 years. The current treatment of neurogenic bladder in patients with SCI involves safely emptying the bladder with a catheter and pursuing further treatments when problems arise.
One common problem encountered is urine incontinence, or leaking between catheterizations, which can cause medical complications including skin breakdown and decreased quality of life. Anticholinergic bladder medications that can help are often prescribed at higher doses in the SCI population and have intolerable side effects that contribute to low compliance. Furthermore, emerging evidence suggests that increased amounts of this category of medication is associated with dementia. Other treatment strategies, including newer medications and bladder injections, are often too expensive or otherwise difficult to obtain. There is a need for alternative treatment strategies that are affordable and can be obtained anywhere.
We investigated whether neuromodulation devices could overcome these obstacles in the treatment of neurogenic bladder in SCI. Neuromodulation devices alter nerve activity by applying electric stimulation to target nerves. We chose to test transcutaneous tibial nerve stimulation (TTNS) as a feasible neuromodulation device with the potential to be initiated during inpatient rehabilitation and continued at home. For 30 minutes, the sticky electrodes are applied to the ankle, which stimulates the tibial nerve. The presumed mechanism is that the tibial nerve enters the spinal cord at the same level as the bladder nerves and interrupts the reflexive overactivity that occurs due to SCI, allowing the bladder to relax, store urine better and reduce episodes of incontinence.
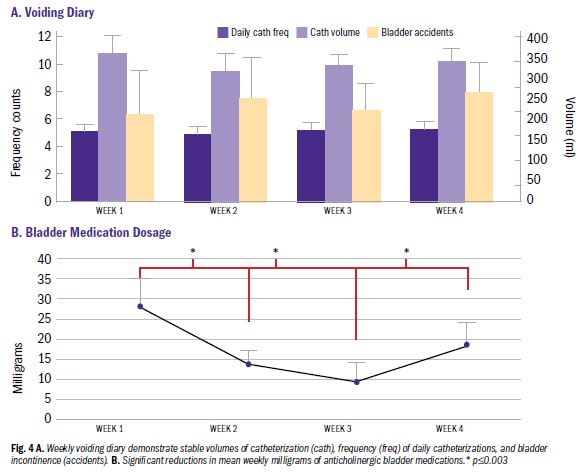
With these encouraging findings, the next steps include performing a randomized control trial of TTNS in the home setting, and ultimately performing a trial beginning during acute injury and continuing to home, measuring long-term outcomes. We anticipate that this line of research will reveal a novel, safe, feasible and cost-effective treatment strategy to improve neurogenic bladder outcomes. Furthermore, this treatment strategy can be performed by the patient (or caregiver) and possibly reduce common urinary morbidities, improve quality of life and reduce medication use that has detrimental side effects.
1 Stampas A, Korupolu R, Zhu L, Smith CP, Gustafson K. Safety, Feasibility, and Efficacy of Transcutaneous Tibial Nerve Stimulation in Acute Spinal Cord Injury Neurogenic Bladder: A Randomized Control Pilot Trial. Neuromodulation. 2018 Oct 3; doi.org/10.1111/ner.12855.
Dr. Argyrios Stampas is research director of the TIRR Memorial Hermann Spinal Cord Injury Medicine Program and a clinical assistant professor in the department of Physical Medicine and Rehabilitation at McGovern Medical School at UTHealth.
